Type 1 Diabetes Implant Device Could Replace Insulin Injections
Researchers at Massachusetts Institute of Technology are developing an innovative type 1 diabetes implant device designed to deliver insulin from within the body—potentially changing how type 1 diabetes is managed.
A New Approach to Managing Blood Sugar
For most people living with diabetes, daily life involves constant blood sugar monitoring and multiple insulin injections to prevent dangerous spikes.
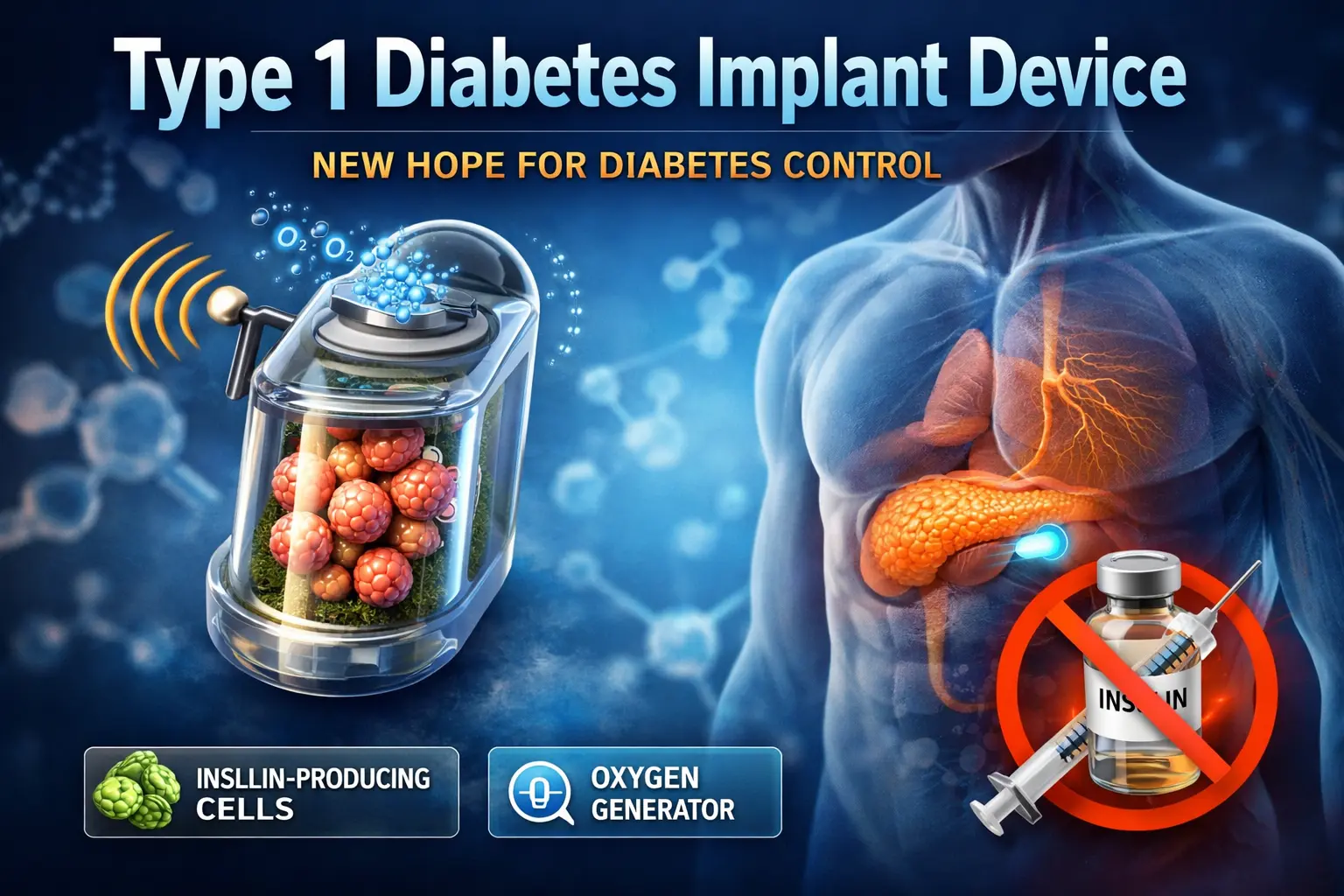
MIT researchers are working on a different solution: an implantable device filled with insulin-producing cells, known as pancreatic islet cells. The device is designed to function inside the body, producing insulin automatically when needed.
How the Implantable Device Works
The device uses a protective encapsulation system that shields the islet cells from the immune system, helping prevent rejection. At the same time, it includes a built-in oxygen generator to keep the cells alive and functioning properly.
This combination addresses one of the biggest challenges in cell-based therapies—keeping transplanted cells both protected and healthy.
Promising Results in Early Studies
In a recent study published in the journal Device, the MIT team demonstrated that the encapsulated islet cells could survive in the body for at least 90 days.
Tests in mice showed that:
- The implanted cells remained functional
- They produced insulin
- They helped maintain stable blood sugar levels
While the treatment did not fully reverse diabetes, it did provide meaningful glucose control.
Eliminating the Need for Immunosuppressive Drugs
“Islet cell therapy can be a transformative treatment for patients. However, current methods also require immune suppression, which for some people can be really debilitating,” said Daniel Anderson, senior author of the study.
“Our goal is to find a way to give patients the benefit of cell therapy without the need for immune suppression.”
The study also included contributions from Siddharth Krishnan, Matthew Bochenek, and Robert Langer.
The Challenge With Traditional Islet Transplants
Islet cell transplantation has already shown success in treating diabetes. These cells are typically sourced from donors or created using stem cell technology.
However, patients must take immunosuppressive drugs to prevent rejection, which can lead to serious side effects.
Encapsulation devices have been explored as an alternative, but they often limit oxygen flow—making it difficult for the cells to survive long-term.
The Breakthrough: Built-In Oxygen Generation
To solve this, the MIT team developed a device with an internal oxygen generator.
This system uses a proton-exchange membrane to split water vapor naturally found in the body into hydrogen and oxygen. The hydrogen diffuses away, while the oxygen is stored and delivered directly to the islet cells through a thin, permeable membrane.
Earlier versions of the device kept cells alive for about a month in mice.
“A month is a good timeframe in that it shows basic proof-of-concept. But from a translational standpoint, it’s important to show that you can go quite a bit longer than that,” Krishnan explained.
Improvements That Extended Cell Survival
In the latest version, researchers improved the device by:
- Making it more waterproof
- Increasing resistance to cracking
- Upgrading electronics to boost oxygen generation
The device is powered wirelessly through an external antenna placed on the skin, which transmits energy to the implant.
These refinements allowed the system to function for at least 90 days in animal models.
Stem Cells Could Provide Unlimited Supply
The researchers also tested islet cells derived from induced pluripotent stem cells, which could eventually provide an unlimited supply of insulin-producing cells.
While the implants did not completely cure diabetes, they significantly improved blood sugar control.
“We’re hoping that in the future, if we can give the cells a little bit longer to fully mature, that they’ll secrete even more insulin to better regulate diabetes in the animals,” said Bochenek.
Here’s more breakthroughs in pancreatic cell replacement!
What Comes Next
The next goal is to extend the lifespan of the implant to two years or more.
“Long-term survival of the islets is an important goal,” Anderson said. “The cells, if they’re in the right environment, seem to be able to survive for a long time.”
Beyond Diabetes: A Platform for Treating Disease
The team is also exploring whether this technology could be used to deliver other therapeutic proteins, including:
- Antibodies
- Enzymes
- Clotting factors
“We think that these technologies could provide a long-term way to treat human disease by making drugs in the body instead of outside of the body,” Anderson said.
“There are many protein therapies where patients must receive repeated, lengthy infusions. We think it may be possible to create a device that could continuously create protein therapeutics on demand.”
Bottom Line
This implantable device represents a promising step toward long-term, self-regulating insulin delivery without the need for daily injections or immunosuppressive drugs.
While still in early stages, the technology could reshape how type 1 diabetes—and potentially other diseases—are treated in the future.
Explore more tools like our A1C Calculator to better understand your blood sugar trends.







Leave a Reply